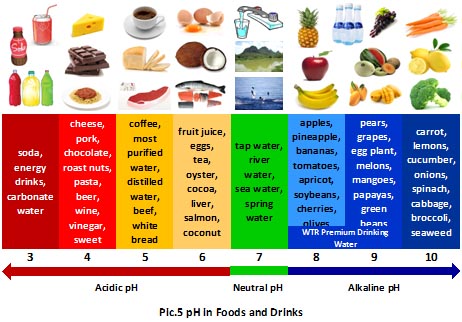
pH |
pH is the measure of the hydronium concentration in a solution. Scientist have come up with a "key" to telling whether something is a basic or acid. They call is the pH scale. The scale is in a range of 0-14. Anything higher than 7 is a base and anything lower than 7 is an acid. Anything that's a 7 is neutral. To figure out the balance of a certain substance, you would use pH= -log(H^+), H representing the hydronium. It will look like pH=-log(1x10^4)
|
pOH |
pOH is power of hydroxide ion concentration and is figured out the same way as pH. However, instead of just using hydronium, it uses hydroxide. To figure the pOH, you would use the equation pOH=-log(OH^+).
Now, if you were to already have either the pH or pOH of a substance, all you would need to do is subtract what you have by 14, since pH + pOH=14.
|
|
|